Class 9 Matter In Our Surroundings Notes
Notes Important Questions NCERT Solutions MCQ Quiz – 1 MCQ Quiz – 2Matter In Our Surroundings Notes
Physical Nature of Matter:-
- Matter is made up of particles.
- Particles of matter are very small that we can’t see by our naked eyes.
Characteristics of Particles of Matter:-
- Particles of matter have space between them.
- Particles of matter are continuously moving.
- Particles of matter attract each other.
We can change the state of matter :-
- by changing the temperature.
- by changing the pressure.
Diffusion:-
The intermixing of particles of two different types of matter on their own is called diffusion.
- On increasing the temperature, the kinetic energy of the particles increases and the particles move faster and so diffusion becomes faster.
Boiling Point:- The temperature at which a liquid starts boiling at the atmospheric pressure is known as its boiling point.
Melting Point:- The temperature at which a solid starts melting to become a liquid at the atmospheric pressure is known as its melting point.
- Melting point of ice = 0 oC = 273.16 K
- Boiling point of water = 100 oC = 373.16 K
Interconversion of three States of Matter:-
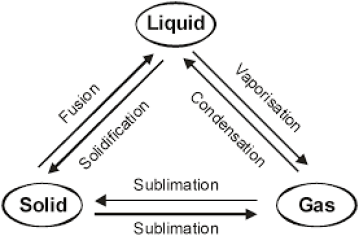
Fusion:- The process of change of solid into liquid state is known as fusion.
Sublimation:- The conversion of solid directly into gaseous state without changing into liquid state (or vice versa) is called sublimation.
- Some sublimable solids are ammonium chloride, camphor, dry ice (solid form of CO2) etc.
Vaporisation:- The conversion of a liquid into gaseous state at its boiling point is called vaporisation.
Latent heat of Fusion:-
The amount of heat energy required to change 1 kg of solid into liquid at atmospheric pressure at its melting point is known as latent heat of fusion.
Latent heat of vaporisation:-
The amount of heat energy required to change 1 kg of liquid into gaseous state at the atmospheric pressure at its boiling point is known as latent heat of vaporisation.
Evaporation:-
The conversion of a liquid into gaseous state at any temperature below its boiling point is called evaporation.
Factors on which rate of evaporation depends are:-
- Surface Area:- On increasing surface area, rate of evaporation also increases.
- Temperature:- On increasing the temperature, rate of evaporation also increases.
- Wind Speed:- With the increase in wind speed, rate of evaporation also increases.
- Humidity:- With the increase in humidity, rate of evaporation decreases.
- During evaporation, the liquid absorbs heat energy from the surroundings to get converted into gaseous state and so, evaporation causes cooling.
Differences Between Vaporisation and Evaporation:-
| S. No. | Evaporation | Vaporisation (Boiling) |
| 1. | The conversion of a liquid into gaseous state at any temperature below its boiling point is called evaporation. | The conversion of a liquid into gaseous state at its boiling point is called vaporisation. |
| 2. | It is a surface phenomenon. | It is a bulk phenomenon. |
| 3. | It is a slow process. | It is a fast process. |
| 4. | It causes cooling. | It does not cause cooling. |
- Now scientists are talking of five states of matter: Solid, Liquid, Gas, Plasma and Bose-Einstein Condensate.
Formulas for interconversion of different units of temperature:-
- oF = (9/5) oC + 32
- oC = (5/9) (oF -32)
- K = oC + 273.16
- oC = K -273.16
- S.I. unit of temperature is Kelvin (K)


Comments