Class 10 Carbon and its Compounds Notes
Notes Previous Years Questions Important QuestionsCarbon And Its Compounds – Notes
Covalent compound:-
The compound formed by the sharing of electrons between the atoms is known as covalent compound.
Difference between covalent compound and ionic/electrovalent compounds:-
| No. | Covalent compounds | Ionic/Electrovalent compounds |
| (i) | Covalent compounds are formed by the sharing of electrons between atoms. | Ionic compounds are formed by the transfer of electrons from an atom to the other atom. |
| (ii) | The force of attraction between the molecules is not very strong. | The force of attraction between the ions is very strong. |
| (iii) | They have low melting point and boiling point. | They have high melting point and boiling point. |
| (iv) | They do not conduct electricity. | They conduct electricity in aqueous or molten state. |
| (v) | They are generally soluble in organic solvent like oil but not in water. | They are generally water soluble. |
Why do carbon not form ionic compounds by gaining or loosing electrons ?
Carbon has four electrons in its outermost shell. So,
- If it gains four electrons forming C4- anion, it would be difficult for the nucleus with six protons to hold on to ten electrons.
- If it loses four electrons forming C4+ cation, it would require a large amount of energy to remove four electrons leaving behind a carbon cation with six protons in its nucleus holding on to just two electrons.
Electron dot structure:-
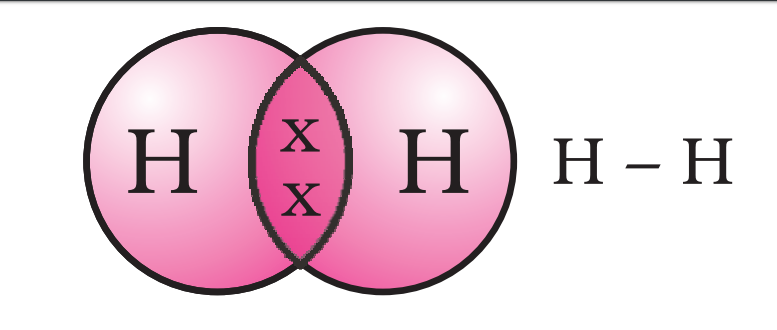
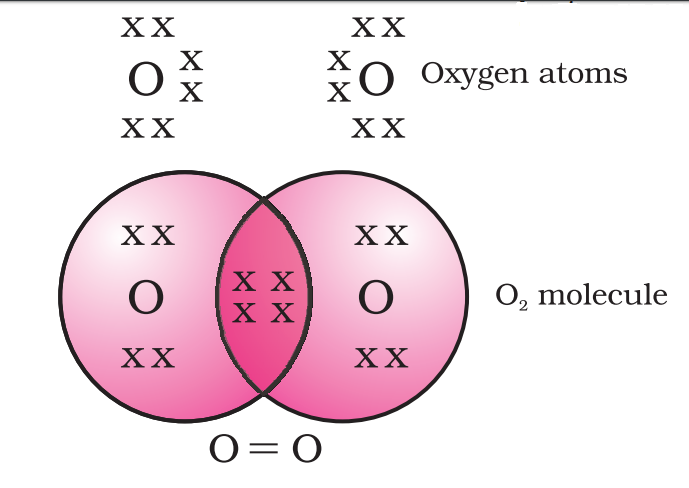
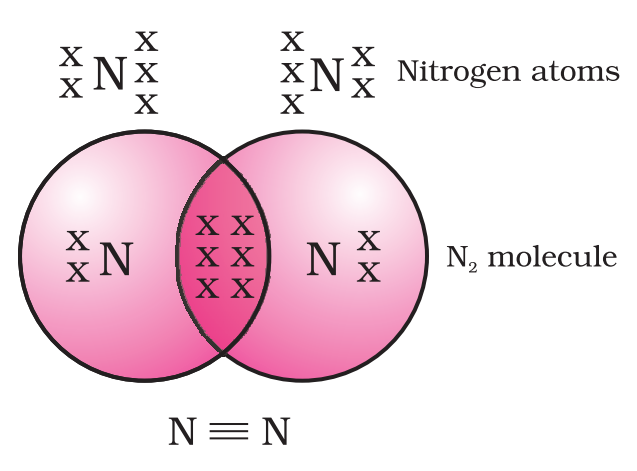
Allotropes:-
The different forms of an element having different physical properties but same chemical properties are called allotropes.
Allotropes of carbon:-
- Graphite
- Diamond
- Fullerene
Graphite:-
- Structure:- Hexagonal array
- It is smooth and slippery and so it is used as lubricant.
- It is a good conductor of electricity.
Diamond:-
- Structure:- Tetrahedral
- It is the hardest substance known.
- Artificial diamond can be synthesized by subjecting pure carbon to very high pressure and temperature.
Fullerene:-
- The first fullerene identified was C-60 which has carbon atoms arranged in the shape of football.
Versatile nature of carbon:-
Carbon forms a lot of compounds due to the following factors:-
- Catenation:- The self linking property of carbon due to which it forms very long chain of carbon compounds and large number of molecules is called catenation.
- Silicon forms compounds with hydrogen having chains of up-to 7 or 8 atoms.
- Tetravalent:- Since carbon has a valency of four so it is capable of bonding with four other atoms of carbon or atoms of other monovalent element. This property is called tetravalent.
- Small size of carbon atom:- Due to its small size, the nucleus of carbon atom can hold the shared paired of electrons strongly. Hence, the bond of carbon are very strong and stable.
Hydrocarbons:-
Carbon compounds which contain only carbon and hydrogen atoms are called hydrocarbons.
Saturated compounds:-
Compounds of carbon having only single bonds between the atoms are called saturated compounds. Ex:- Alkane (CnH2n + 2)
Unsaturated compounds:-
Compounds of carbon having double or triple bonds between the carbon atoms are called unsaturated compounds. Ex:- Alkene (CnH2n), Alkyne (CnH2n – 2).
- Saturated carbon compounds are more stable than unsaturated carbon compounds.
Distinguish between saturated hydrocarbons and unsaturated hydrocarbons:-
When bromine water is added to saturated hydrocarbons then it will have no change in its colour, but when it is added to unsaturated hydrocarbons, its colour will be decolourised (colourless).
Structural isomers:-
The compounds with same molecular formula but different structures are called structural isomers. Ex:-
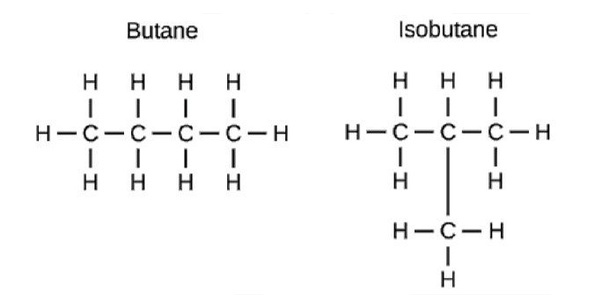
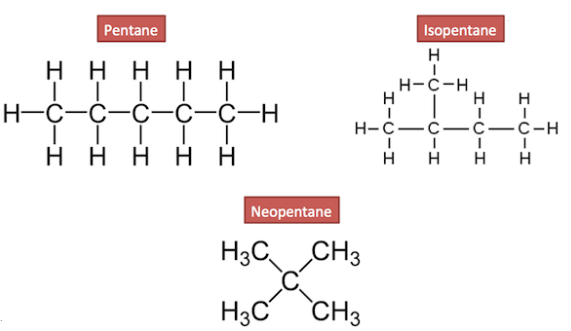
Heteroatom:-
The element replacing hydrogen atom in carbon compound is called heteroatom.
Functional group:-
An atom or a group of atoms which largely determines the properties of carbon compounds is called functional group.
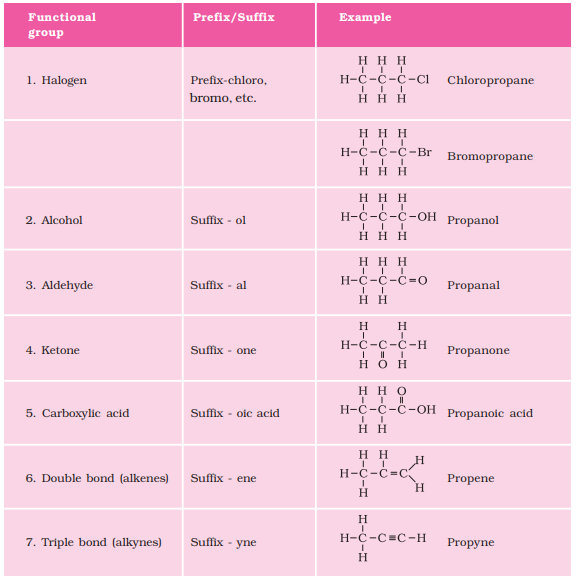
| Heteroatom | Functional Group | Formula | Prefix/Suffix |
| Fluorine/Chlorine/ Bromine/Iodine | Halogen (Fluorine/ Chlorine/ Bromine/Iodine) | – F – Cl – Br – I | fluoro chloro bromo iodo |
| Oxygen | Alcohol | – OH | ol |
| Oxygen | Aldehyde | 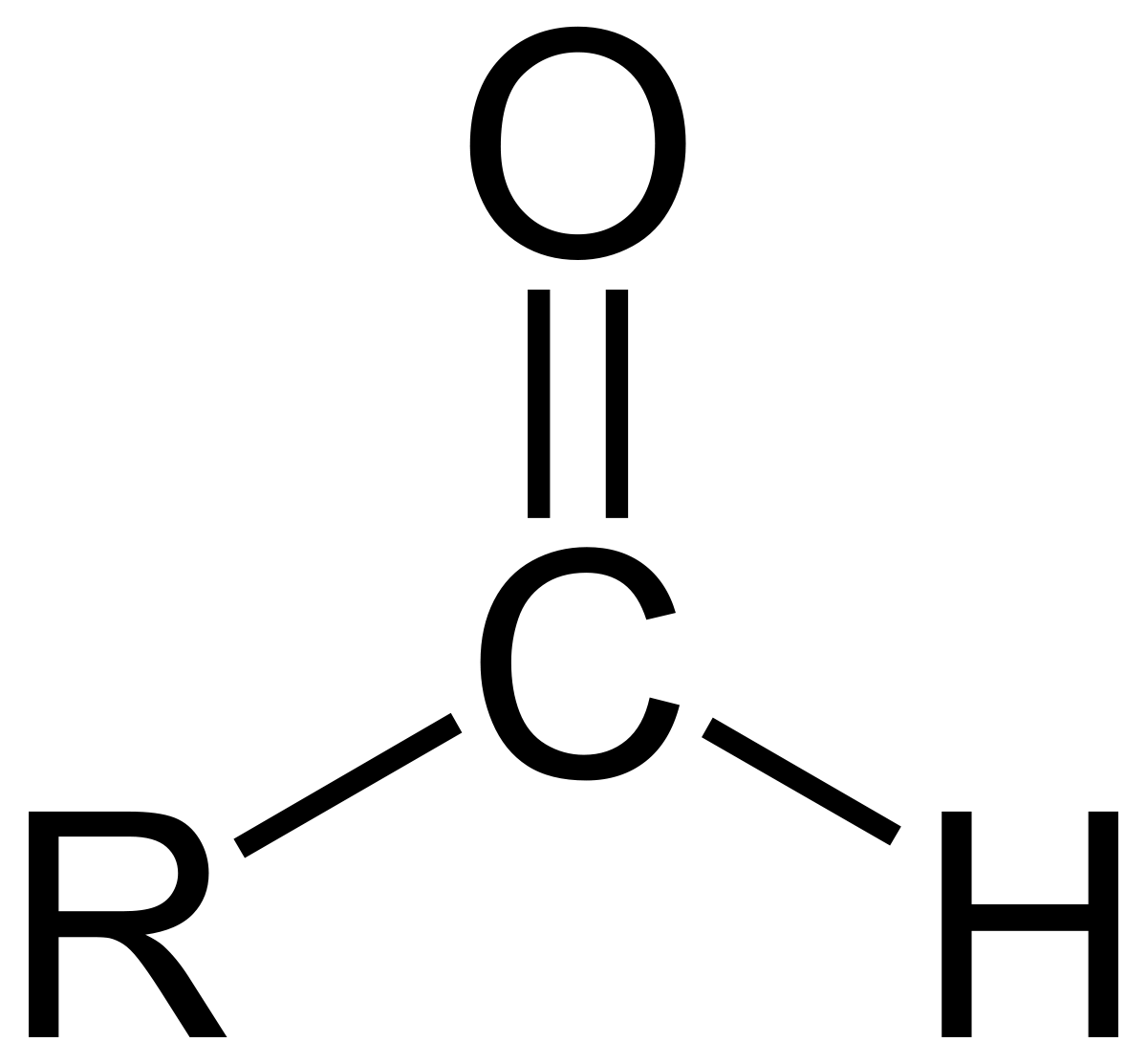 | al |
| Oxygen | Ketone | 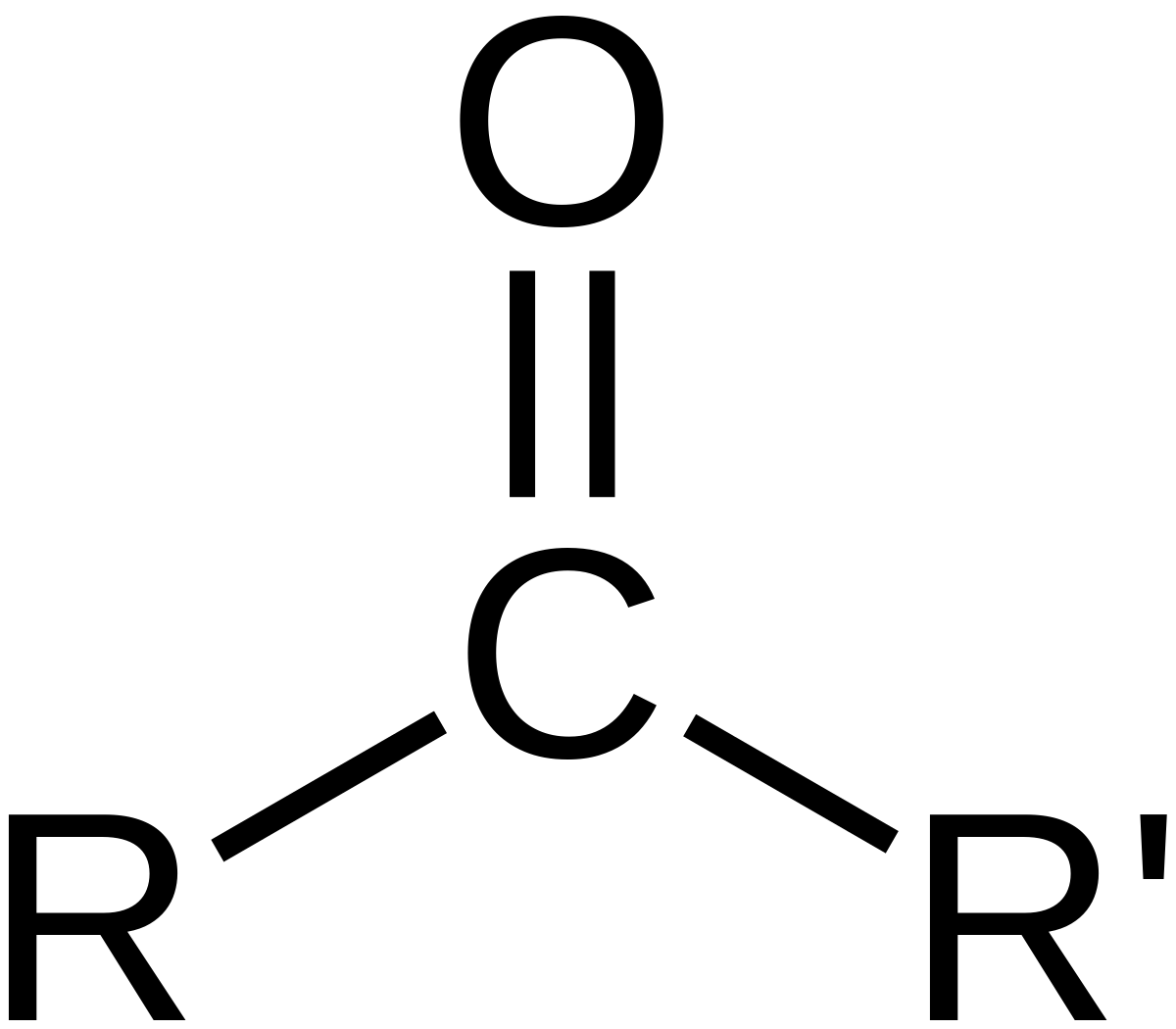 | one |
| Oxygen | Carboxylic acid | 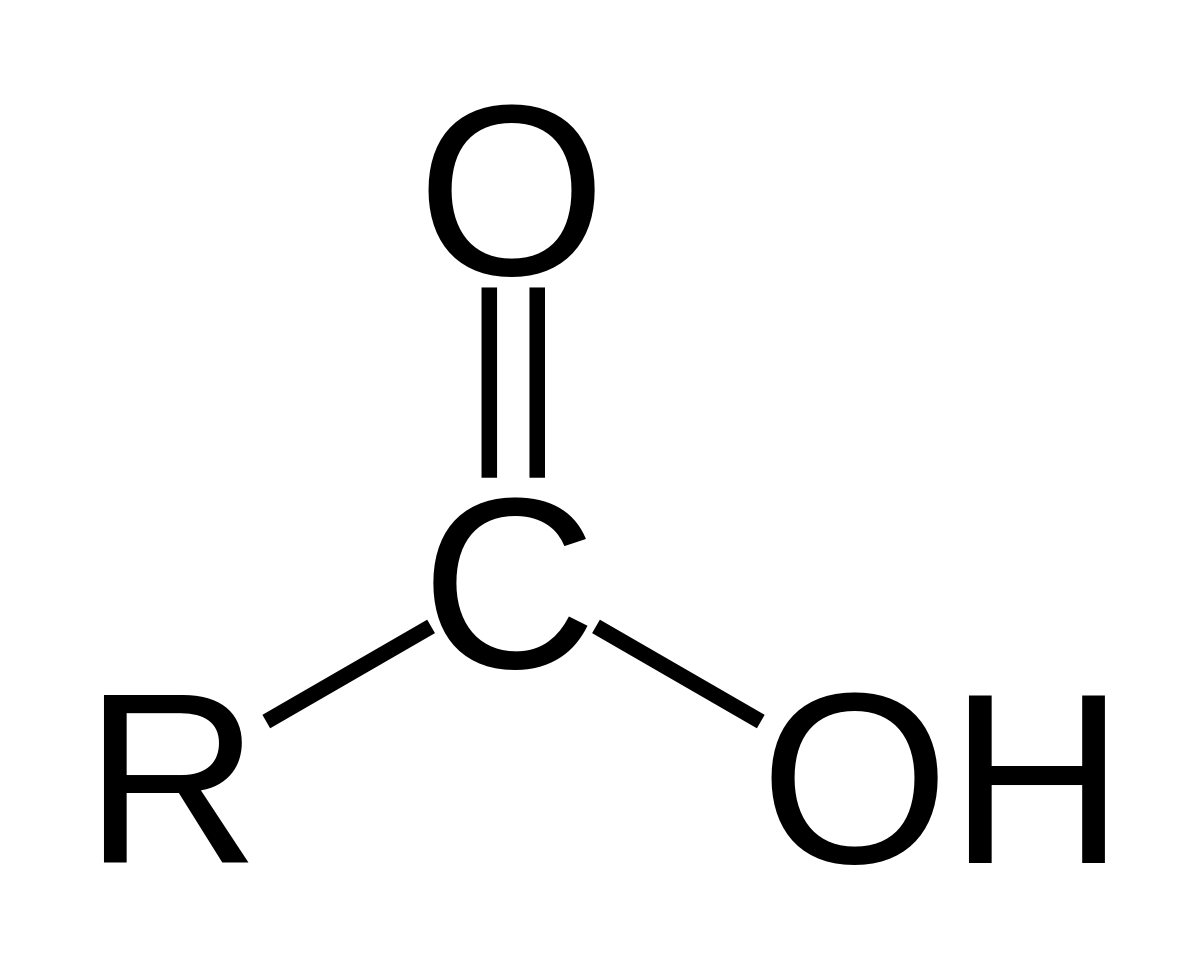 | oic acid |
Homologous series:-
A series of carbon compounds in which the same functional group replaces hydrogen atom in a carbon chain is called homologous series. Ex:-
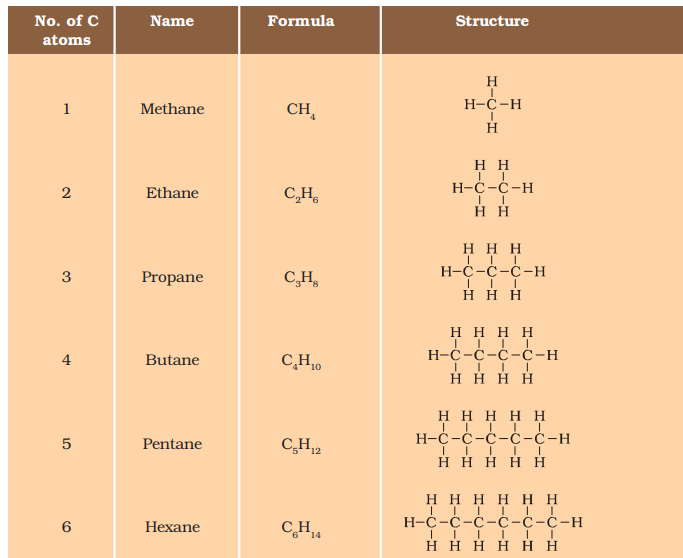
Alkane:-
CH4 (Methane), C2H6 (Ethane), C3H8 (Propane), C4H10 (Butane), C5H12 (Pentane), ………….
Alkanol (Alcohol):-
CH3OH (Methanol), C2H5OH (Ethanol), C3H7OH (Propanol), C4H9OH (Butanol),…….
- The two successive members of a homologous series differ by – CH2 unit and by molecular mass of 14 unit.
Nomenclature of functional groups:-
Chemical properties of carbon compounds:-
Combustion:-
The burning of carbon compounds in the presence of oxygen is called combustion.
- C + O2 → CO2 + heat and light
- CH4 + 2O2 → CO2 + 2H2O+ heat and light
- On combustion, saturated hydrocarbons generally give a clean blue flame while unsaturated hydrocarbons give yellow flame with lots of smoke.
- Saturated hydrocarbons give sooty flame (yellow flame with black smoke) in limited supply of oxygen.
Oxidation:-
When alcohols are heated in the presence of alkaline KMnO4 (potassium permanganate) or acidified K2Cr2O7 (potassium dichromate), they get converted to carboxylic acid.

- Here, alkaline KMnO4 and acidified K2Cr2O7 are oxidising agents.
Addition Reaction:-
The addition of hydrogen to unsaturated hydrocarbons in the presence of nickel or palladium catalyst to give saturated hydrocarbons is known as addition reaction.
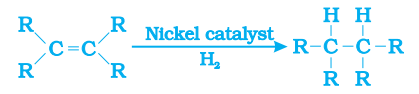
- This reaction is used in the hydrogenation of vegetable oil using nickel catalyst.
- Animal fats generally contain saturated fatty acids which are harmful for our health.
- Vegetable oils generally contain unsaturated fatty acids which are good for our health.
Substitution reaction :-
The reaction in which chlorine is added to hydrocarbons in the presence of sunlight is called substitution reaction. Ex:-
CH4 + Cl2 → CH3Cl + HCl (in the presence of sunlight)
- Melting point and boiling point increase with increasing molecular mass.
- Solubility in a particular solvent also increases with increasing molecular mass.
Ethanol (CH3CH2OH):-
- It is liquid at room temperature.
- It is used to prepare alcoholic drinks.
- It is a good solvent.
- It is used in medicines such as tincture of iodine, cough syrups and many tonics.
- It is soluble in water.
- Melting point is 156 K and boiling point is 351 K.
Reactions of ethanol:-
- Reaction with sodium:-
2Na + 2CH3CH2OH → 2CH3CH2ONa + H2
➢ CH3CH2ONa ⇒ Sodium ethoxide - Reaction to give unsaturated hydrocarbons:-

➢ Hot conc. H2SO4 ⇒ Dehydrating agent
Ethanoic acid (CH3COOH):-
- Common name:- Acetic acid
- Melting point of pure ethanoic acid is 290 K (17 °C)
- It is also called glacial acetic acid because it freezes in winter.
- Boiling point is 391 K.
- It is a weak acid because it does not ionises completely.
- Vinegar:- 5-8 % solution of acetic acid in water is called vinegar.
- Vinegar is used as preservative in pickles.
Reactions of ethanoic acid:-
- Esterification reaction:- The reaction of an acid and an alcohol to give an ester in the presence of an acid catalyst is called esterification reaction.
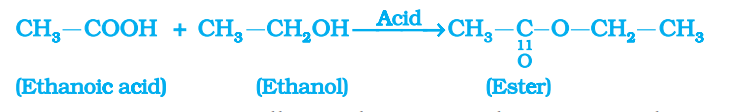
- Properties of ester:-
➢ Esters are sweet smelling substances.
➢ Esters are used in perfumes and as flavoring agents.
Saponification reaction:- The reaction of an ester with an acid or a base to give back the alcohol and carboxylic acid is known as saponification reaction.
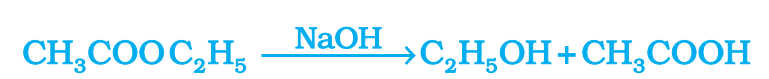
➢ Saponification reaction is used in the preparation of soaps.
- Reaction of ethanoic aicd with a base:-
CH3COOH + NaOH → CH3COONa + H2O
CH3COONa ⇒ Sodium ethanoate (Sodium acetate)
- Reaction of ethanoic acid with carbonates or hydrogen carbonates:-
2CH3COOH + Na2CO3 → 2CH3COONa + H2O + CO2
CH3COOH + NaHCO3 → CH3COONa + H2O + CO2
Soaps and detergents:-
Soaps:-
Soap molecules are sodium or potassium salts of long chain carboxylic acid.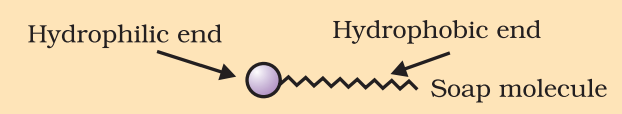
Scum:- The insoluble precipitate formed by the reaction of soap molecules with calcium and magnesium salts present in hard water is called scum.
Hard water:- Water containing calcium or magnesium salts is called hard water.
Detergents:-
- Detergents are ammonium or sulphonate salts of long chain carboxylic acid.
- Detergent does not form insoluble precipitate in hard water. So, It remain effective in hard water also.
- Detergents are used to make shampoo and products for cleaning clothes.
Cleansing action of soap:-
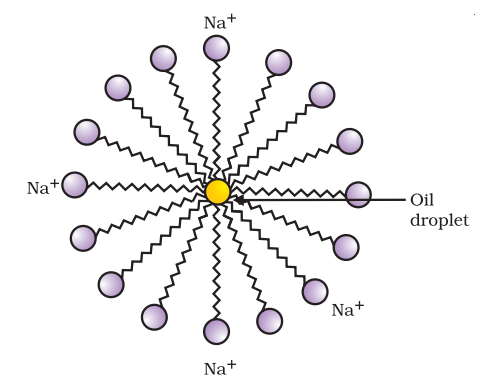
The dirt present on the clothes is oily in nature. When we apply soap on the clothes, the hydrophobic (water repelling) end of soap molecules get attached with dirt and the hydrophilic (water loving) end move away from the dirt forming a structure called micelle. The hydrophillic end gets attached to water and the dirt is removed along with water and micelle.
Micelle:-


Comments