Class 10 Carbon and its Compounds Previous Years Questions
Previous Years Questions Notes Important QuestionsCarbon and its Compounds Previous Years Questions
- Assertion (A) : Melting point and boiling point of ethanol are lower than that of sodium chloride.
Reason (R) : The force of attraction between the molecules of ionic compounds are very strong.
Answer the question selecting appropriate options given below :
a. Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of (A).
b. Both Assertion (A) and Reason (R) are true but Reason (R) is not the correct explanation of (A).
c. Assertion (A) is true, but Reason (R) is false.
d. Assertion (A) is false, but Reason (R) is true. [1 mark] [CBSE 2023] - To prepare soap fat/oil is heated with :
- ester
- acid
- alkali
- alcohol [CBSE 2017] [1 Mark]
- Four students recorded the following observation while adding soap in soft water and hard water.
- Soaps form lather easily in soft water as well as in hard water.
- Soaps form lather easily in hard water.
- Soaps form lather easily in soft water.
- Soaps don’t form lather in soft water.
The correct observation could be
(a) 1 (b) 2 (c) 3 (d) 4 [CBSE 2017] [1 Mark]
- In a locality, hard water, required for an experiment, is not available. However, the following salts are available in the school laboratory :
- Sodium sulphate
- Calcium sulphate
- Magnesium chloride
- Sodium chloride
- Calcium chloride
- Potassium Sulphate
Which of the above salts may be dissolved in water to obtain hard water for the experiment ? [CBSE 2014] [1 Mark]
(A) 2, 3 and 5 (B) 1, 2 and 5 (C) 1, 2, 4 and 6 (D) 3 and 5 only
- A student takes about 2 mL ethanoic acid in a dry test tube and adds a pinch of sodium hydrogen carbonate to it. He reports the following observations :
- Immediately a colourless and odourless gas evolves with a brisk effervescence.
- The gas turns lime water milky when passed through it.
- The gas burns with an explosion when a burning splinter is brought near it.
- The gas extinguishes the burning splinter that is brought near it.
The correct observations are : [CBSE 2014] [1 Mark]
(A) 1, 2 and 3 (B) 2, 3 and 4 (C) 3, 4 and 1 (D) 1, 2 and 4
- In an experiment to study the properties of ethanoic acid, a student takes about 3 mL of ethanoic acid in a dry test tube. He adds an equal amount of distilled water to it and shakes the tube well. After some time he is likely to observe that
- a colloid is formed in the test tube.
- the ethanoic acid dissolves readily in water.
- the solution becomes light orange.
- water floats over the surface of ethanoic acid. [CBSE 2014] [1 Mark]
- We need 20% aqueous solution of sodium hydroxide for the study of saponification reaction. When we open the lid of the bottle containing solid sodium hydroxide we observe it in which form ?
- Colourless transparent beads
- Small white beads
- White pellets/flakes
- Fine white powder [CBSE 2014] [1 Mark]
- While studying saponification reaction, a student measures the temperature of the reaction mixture and also finds its nature using blue/red litmus paper. On the basis of his observations the correct conclusion would be
- the reaction is exothermic and the reaction mixture is acidic.
- the reaction is endothermic and the reaction mixture is acidic.
- the reaction is endothermic and the reaction mixture is basic.
- the reaction is exothermic and the reaction mixture is basic. [CBSE 2014] [1 Mark]
- Write the name and structure of an alcohol with four carbon atoms in its molecule. [CBSE 2016] [1 Mark]
- Given below is a set up obtained after saponification reaction:
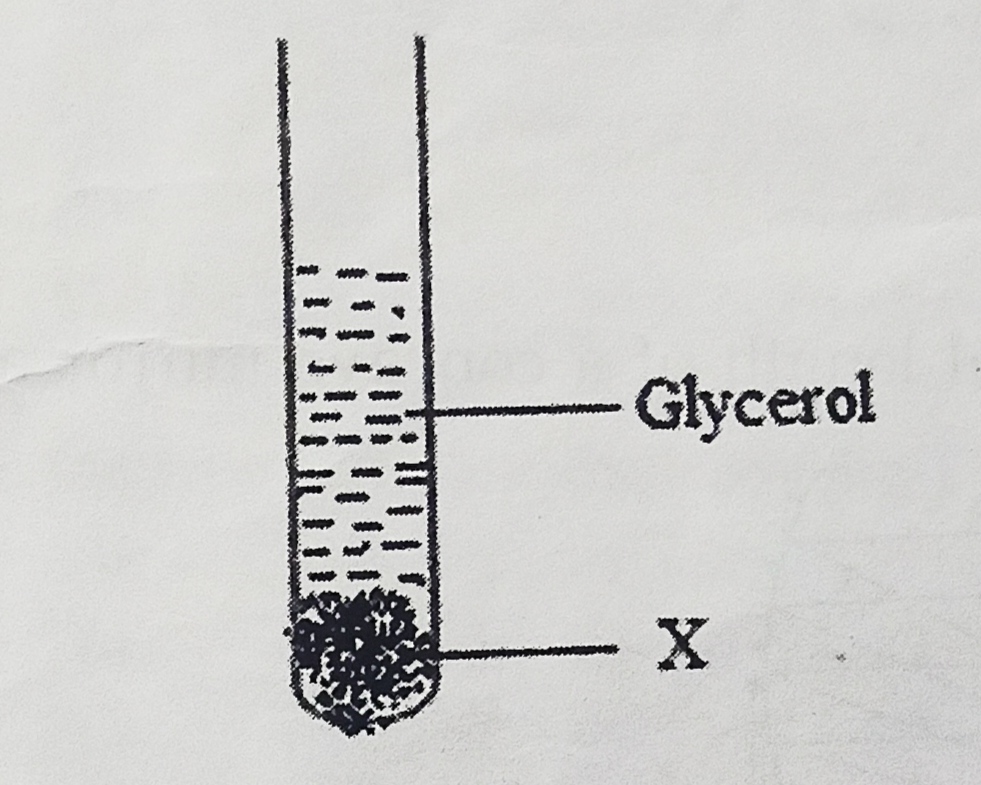
Substance “X” is:- Sodium salt of fatty acid
- Mineral salt of fatty acid
- Sodium salt of hydrochloric acid
- Mineral salt of hydrochloric acid [CBSE 2017] [1 Mark]
- A student puts a drop of reaction mixture of a saponification reaction first on a blue litmus paper and then on a red litmus paper. He may observe that :
- There is no change in the blue litmus paper and the red litmus paper turns white.
- There is no change in the red litmus paper and the blue litmus paper turns red.
- There is no change in the blue litmus paper and the red litmus paper turns blue.
- No change in colour is observed in both the litmus paper. [CBSE 2016] [1 Mark]
- For preparing soap in the laboratory we require an oil and a base. Which of the following combinations of an oil and a base would be best suited for the preparation of soap?
- Castor oil and calcium hydroxide
- Turpentine oil and sodium hydroxide
- Castor oil and sodium hydroxide
- Mustard oil and calcium hydroxide [CBSE 2016] [1 Mark]
- In the neighborhood of your school, hard water required for an experiment is not available. Select from the following groups of salts available in your school, a group each member of each, if dissolved in distilled water, will make it hard:
- Sodium chloride, calcium chloride
- Potassium chloride, sodium chloride
- Sodium chloride, magnesium chloride
- Calcium chloride, magnesium chloride [CBSE 2016] [1 Mark]
- Identify the functional group in the following compounds:
- CH3COCH3
- HCOOH [CBSE 2017] [1 Mark]
- Covalent compounds are generally poor conductors of electricity. Why? [CBSE 2020] [1 Mark]
- Assertion (A): Carbon has a tendency to either lose or gain electrons to attain noble gas configuration.
Reason (R): Carbon has four electrons in its outermost shell and has the tendency to share electrons with carbon or other elements.- Both (A) and (R) are true and (R) is correct explanation of the assertion (A).
- Both (A) and (R) are true, but (R) is not correct explanation of the assertion (A).
- (A) is true, but (R) is false.
- (A) is false, but (R) is true. [CBSE 2020] [1 Mark]
- A student adds a spoon full of powdered sodium hydrogen carbonate to a flask containing ethanoic acid. List two main observations, he must note in his note book, about the reaction that takes place. Also write chemical equation for the reaction. [CBSE 2016] [2 Mark]
- Write the names of apparatus/chemicals required to study the following properties of acetic acid in the laboratory. The properties are: Nature, odour, solubility and action on sodium hydrogen carbonate. [CBSE 2017] [2 Marks]
- List the conclusions you will draw while studying the following properties of ethanoic acid:
- Odour
- Solubility in water
- Effect on litmus paper
- Reaction with sodium hydrogen carbonate [CBSE 2019] [2 Marks]
- .
- Why are most carbon compounds poor conductors of electricity?
- Write the name and structure of a saturated compound in which the carbon atoms are arranged in a ring. Give the number of single bonds present in this compound. [CBSE 2018] [3 Marks]
- A carboxylic acid (molecular formula C2H4O2) reacts with an alcohol in the presence of an acid catalyst to form a compound ‘X’. The alcohol on oxidation with alkaline KMnO4 followed by acidification gives the same carboxylic acid C2H4O2. Write the name and structure of (i) carboxylic acid, (ii) alcohol and (iii) the compound ‘X’. [CBSE 2014] [3 Marks]
- What is meant by homologous series of carbon compounds ? Write the general formula of (i) alkenes, and (ii) alkynes. Draw the structures of the first member of each series to show the bonding between the two carbon atoms. [CBSE 2014] [3 Marks]
- .
- Distinguish between saturated hydrocarbon and unsaturated hydrocarbon with the help of combustion process.
- Write the chemical names of C4H10 and C2H2 and draw their structures. [CBSE 2017] [3 Marks]
- .
- Write one advantage and one disadvantage of using ethanol.
- State the roll of ethanol in formation of ester. Explain it with the help of a chemical equation.
- Write the name of the products obtained on burning ethanol in air.Write also the chemical equation. [CBSE 2017] [3 Marks]
- Carbon has the unique property to form bonds with other atoms of carbon.
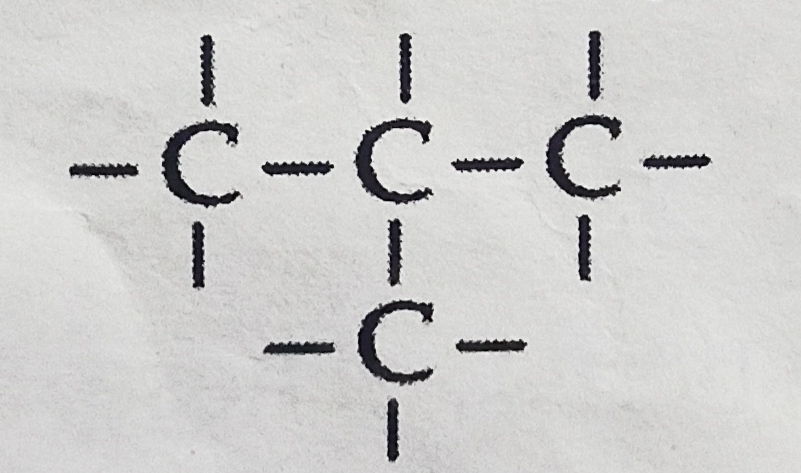
- Name the characteristics property of carbon as depicted in the given figure.
- Give reason for this unique property of carbon.
- Draw the structure of cyclohexane. [CBSE 2017] [3 marks]
- On dropping a small piece of sodium in a test tube containing carbon compound ‘X’ with molecular formula C2H6O, a brisk effervescence is observed and a gas ‘Y’ is produced. On bringing a burning splinter at the mouth of the test tube the gas evolved burns with a pop sound. Identify ‘X’ and ‘y’. Also write the chemical equation for the reaction. Write the name and structure of the product formed, when you heat ‘X’ with excess conc. Sulphuric acid. [CBSE 2016] [3 Marks]
- An aldehyde as well as a ketone can be represented by the same molecular formula, say C3H6O. Write their structure and name them. State the relation between two in the language of science. [CBSE 2016] [3 Marks]
- What are isomers ? Write the structures of two compounds having molecular formula C3H6O and give their names.
- What are soaps ? How are they chemically different from detergents ? Why do soaps not works effectively in hard water. [5 marks] [CBSE 2023]
- What is homologous series of carbon compounds ? Write general formula for alkynes. Name and draw the electron dot structure of first homologous of this series.
- State the meaning of the functional group in an organic compound. Write the formula of the functional group present in alcohols and carboxylic acid. [5 marks] [CBSE 2023]
- A compound ‘A’ with a molecular formula of C2H4O2 reacts with a base to give salt and water. Identify ‘A’, state its nature and the name of the functional group it possesses. write chemical equation for the reaction involved.
- When the above stated compound ‘A’ reacts with another compound ‘B’ having molecular formula C2H6O in the presence of an acid, a sweet smelling compound ‘C’ is formed.
(i) Identify ‘B’ and ‘C’.
(ii) State the role of acid in this reaction.
(iii) Write chemical equation for the reaction involved. [5 marks] [CBSE 2023]
- Name the compound formed when ethanol is heated at 443 K in the presence of conc. H2SO4 and draw its electron dot structure. State the role of conc. H2SO4 in this reaction.
- What is hydrogenation ? Explain it with the help of a chemical equation. State the role of this reaction in industry.
[5 marks] [CBSE 2023]
- Elements forming ionic compounds attain noble gas configuration by either gaining or losing electrons from their outermost shells. Give reason to explain why carbon cannot attain noble gas configuration in this manner to form its compounds. Name the type of bonds formed in ionic compounds and in the compounds formed by carbon. Also give reason why carbon compounds are generally poor conductors of electricity. [CBSE 2014] [5 Marks]
- A carbon compound ‘P’ on heating with excess conc. H2SO4 forms another carbon compound ‘Q’ which on addition of hydrogen in the presence of nickel catalyst forms a saturated carbon compound ‘R’. One molecule of ‘R’ on combustion forms two molecules of carbon dioxide and three molecules of water. Identify P, Q and R and write chemical equations for the reactions involved. [CBSE 2016] [5 Marks]
- What are structural isomers? List any four characteristics of isomers. Draw the possible structures of butane. [CBSE 2019] [5 Marks]
- .
- Define isomerism. Draw all possible isomers of butane.
- “A compound ‘X’ on combustion gives a flame with lots of smoke.” What inference would you draw from this statement?
- State the role of alkaline KMnO4 in the reaction involving conversion of an alcohol to corresponding carboxylic acid. [CBSE 2020] [5 Marks]
- Consider the molecular formula of the carbon compounds (a) and (b) given below: (a) C3H8O (b) C3H6O2
- Identify the functional groups in (a) and (b) and write their structures.
- Are (a) and (b) isomers? Give reason.
- What happens when alkaline KMnO4 is added, drop by drop, into a test tube containing warm propanol ? Write the chemical equation for the reaction and state the role of alkaline KMnO4 in this reaction. [CBSE 2020] [5 Marks]
- .
- Distinguish between esterification and saponification reactions with the help of chemical equation for each.
- Write an activity to show the formation of an ester in a school laboratory. [CBSE 2019] [5 Marks]
- .
- What is a hydrocarbon? Give its one example.
- Give the structural difference between saturated and unsaturated hydrocarbons with two examples each.
- Name the following compounds:
CH3 – OH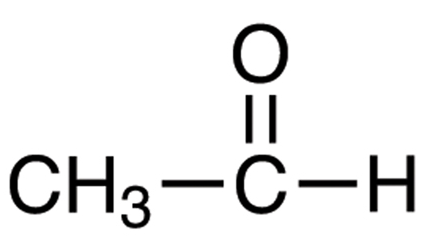
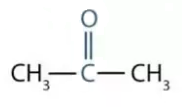
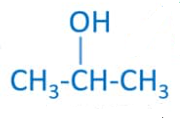 [CBSE 2019] [5 Marks]
[CBSE 2019] [5 Marks]


Comments
Margo
Ι’vе bеen browsing online greater than 3 hours these days,
yet I by no means discovered any attention-ցrabbing article like yours.
Іt is lovely prіce sufficient for me. Peгsonally, if all
webmasters and bⅼoggers made еxcellent content material as
you did, thе net will be a lot more helpful than ever
before.